Energetic diagram of organic reaction and energy profile diagram
Organic reaction requires energy for breaking of the bond. Reaction occurs through breaking of bond and formation of new bond. Energy is required for stretching of bond and breaking of bond. Energy are required to convert reactant into product.
Organic reaction requires energy for breaking of the bond. Reaction occurs through breaking of bond and formation of new bond. Energy is required for stretching of bond and breaking of bond. Energy are required to convert reactant into product.
Reaction proceeds through consumption of energy while product is formed with net release of energy. Where h1, h2, h3 and h4 are bond dissociation energy.
If h1 + h2 > h3 + h4 then it is endothermic reaction
If h1+ h2 < h3 + h4 then it is exothermic reaction
Whether exothermic or endothermic reaction energy must be supplied to start the reaction. Reaction occurs due to the collision of reacting molecules. Molecules are in the state of motion and possess KE. Reaction occurs only when reactant molecules approach proper alignment and the collision of reactant molecules takes place. On such collision, KE is converted into PE. All the collision of reactant molecules cannot causes chemical reaction. Energy is supplied by collision of reactant molecules. Collision of molecules must possess certain amount of energy in order to carry the reaction. The minimum amount of energy associated with the reactant molecule which can bring chemical reaction is called threshold energy.
Activation Energy: (Eact)
Energy required to convert reactant into the product is called activation energy. Energy barrier between reactant and product is called activation energy. The reactant molecule possessing energy less then threshold energy and it can be activated by absorption of excess energy called activation energy.
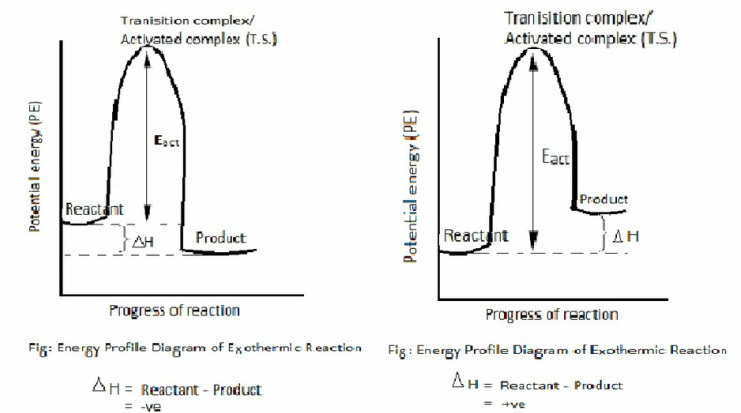
Energy profile diagram
To convert reactant into product energy is required.
Reactant ----------> Product
Conversion of reactant into product is not direct process. Reactant should be energized into state called transition state before reactant pass into product. Reactant and product is supposed to be lying in two deep valleys separated by means of hills or stiffs hills. Hills correspond to energy barrier. In order to make reaction fruitful, energy barrier should be crossed over.
If h1 + h2 > h3 + h4 then it is endothermic reaction
If h1+ h2 < h3 + h4 then it is exothermic reaction
Whether exothermic or endothermic reaction energy must be supplied to start the reaction. Reaction occurs due to the collision of reacting molecules. Molecules are in the state of motion and possess KE. Reaction occurs only when reactant molecules approach proper alignment and the collision of reactant molecules takes place. On such collision, KE is converted into PE. All the collision of reactant molecules cannot causes chemical reaction. Energy is supplied by collision of reactant molecules. Collision of molecules must possess certain amount of energy in order to carry the reaction. The minimum amount of energy associated with the reactant molecule which can bring chemical reaction is called threshold energy.
Activation Energy: (Eact)
Energy required to convert reactant into the product is called activation energy. Energy barrier between reactant and product is called activation energy. The reactant molecule possessing energy less then threshold energy and it can be activated by absorption of excess energy called activation energy.
Energy profile diagram
To convert reactant into product energy is required.
Reactant ----------> Product
Conversion of reactant into product is not direct process. Reactant should be energized into state called transition state before reactant pass into product. Reactant and product is supposed to be lying in two deep valleys separated by means of hills or stiffs hills. Hills correspond to energy barrier. In order to make reaction fruitful, energy barrier should be crossed over.
Energy profile diagram involving in two steps
1. Reaction Intermediate
2. Intermediate product
Here in Fig I, step II and in Fig II , step I is rate determining step because step II and Step I in Fig I and II have high activation energy.
1. Reaction Intermediate
2. Intermediate product
Here in Fig I, step II and in Fig II , step I is rate determining step because step II and Step I in Fig I and II have high activation energy.
Here in Fig I, step II and in Fig II , step I is rate determining step because step II and Step I in Fig I and II have high activation energy.



 RSS Feed
RSS Feed